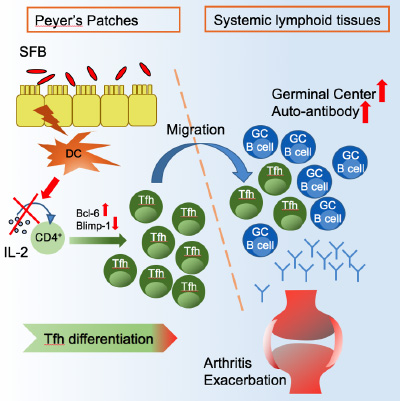
 Rheumatoid arthritis (RA) is an important health problem. It is an autoimmune disease causing chronic inflammation and pain of the joints that affects around 1% of the world's population. The low concordance rate of RA in monozygotic twins (15%) compared to other autoimmune disease such as type I diabetes (~50%) suggests that environmental factors likely play a crucial role in the etiopathogenesis of RA. Our lab research focuses on my long-standing interest in microbes and autoimmune diseases. Imbalance of gut microbiota acts as an environmental cue, triggering many diseases including those located systemically (outside the gut; e.g. rheumatoid arthritis, type 1 diabetes, allergies, etc.). We are especially interested in how commensal bacteria residing in the gut impact autoimmune arthritis. We hypothesize that gut microbiota drive gut-distal (non-gut) autoimmune disease by modulating the mucosal immune response. We have established a new system to examine commensal-host interactions by manipulating the status of the commensal, segmented filamentous bacteria (SFB), in K/BxN arthritis model housed in specific pathogen free (SPF) facility. The new SFB/SPF approach is more physiologically relevant than our prior SFB/germ free (GF) model because commensals normally function in the presence of other commensals, and exert their influence on immunocompetent (e.g. SPF) rather than immunocompromised (e.g. GF) individuals. Notably, our results confirmed the hypothesis that a “benign” species of gut microflora can turn aggressive, acting as an environmental trigger for autoimmune arthritis in a genetically-predisposed mouse model (Teng et al., Immunity 2016). Our recent study reveals a novel mechanism by which a change in a single type of gut commensal, segmented filamentous bacteria (SFB), can trigger systemic joint disease by inducing Peyer’s patch Tfh cells, which then leave the gut and migrate into the periphery causing joint pathology (Fig 1). Though RA classically involves joint disease, it can also affect other organs. Pulmonary complications, which are common extra-articular manifestations, account for 20% of mortality in RA. We have recently branched out to study the gut-lung axis: how gut microbiota affect autoimmunity or infections in the lung. Our research will provide tremendous insight into understanding mucosal-related diseases as well as determining the initiation site of environmentally-triggered systemic diseases.
Rheumatoid arthritis (RA) is an important health problem. It is an autoimmune disease causing chronic inflammation and pain of the joints that affects around 1% of the world's population. The low concordance rate of RA in monozygotic twins (15%) compared to other autoimmune disease such as type I diabetes (~50%) suggests that environmental factors likely play a crucial role in the etiopathogenesis of RA. Our lab research focuses on my long-standing interest in microbes and autoimmune diseases. Imbalance of gut microbiota acts as an environmental cue, triggering many diseases including those located systemically (outside the gut; e.g. rheumatoid arthritis, type 1 diabetes, allergies, etc.). We are especially interested in how commensal bacteria residing in the gut impact autoimmune arthritis. We hypothesize that gut microbiota drive gut-distal (non-gut) autoimmune disease by modulating the mucosal immune response. We have established a new system to examine commensal-host interactions by manipulating the status of the commensal, segmented filamentous bacteria (SFB), in K/BxN arthritis model housed in specific pathogen free (SPF) facility. The new SFB/SPF approach is more physiologically relevant than our prior SFB/germ free (GF) model because commensals normally function in the presence of other commensals, and exert their influence on immunocompetent (e.g. SPF) rather than immunocompromised (e.g. GF) individuals. Notably, our results confirmed the hypothesis that a “benign” species of gut microflora can turn aggressive, acting as an environmental trigger for autoimmune arthritis in a genetically-predisposed mouse model (Teng et al., Immunity 2016). Our recent study reveals a novel mechanism by which a change in a single type of gut commensal, segmented filamentous bacteria (SFB), can trigger systemic joint disease by inducing Peyer’s patch Tfh cells, which then leave the gut and migrate into the periphery causing joint pathology (Fig 1). Though RA classically involves joint disease, it can also affect other organs. Pulmonary complications, which are common extra-articular manifestations, account for 20% of mortality in RA. We have recently branched out to study the gut-lung axis: how gut microbiota affect autoimmunity or infections in the lung. Our research will provide tremendous insight into understanding mucosal-related diseases as well as determining the initiation site of environmentally-triggered systemic diseases.
The practical experimental techniques involved in the Wu lab include transgenic and (conditional) knock out mouse models, flow cytometry, microarrays and accompanying computational analysis, T and B cell adoptive transfer systems, ex vivo and in vitro functional studies of systemic and mucosal immune compartments, microbial profiling, and cell signaling assays.

